

The ISOMED product range of video isolation amplifiers has been established in the market in the field of patient safety for many years. In 2011, Ankrit Technologies took over the production and sales rights and has been looking after the extensive customer base since then. We supply specialist retailers, IT system houses, manufacturers of diagnostic devices, hospitals and GP practices who use their medical devices responsibly with our isolation amplifiers.









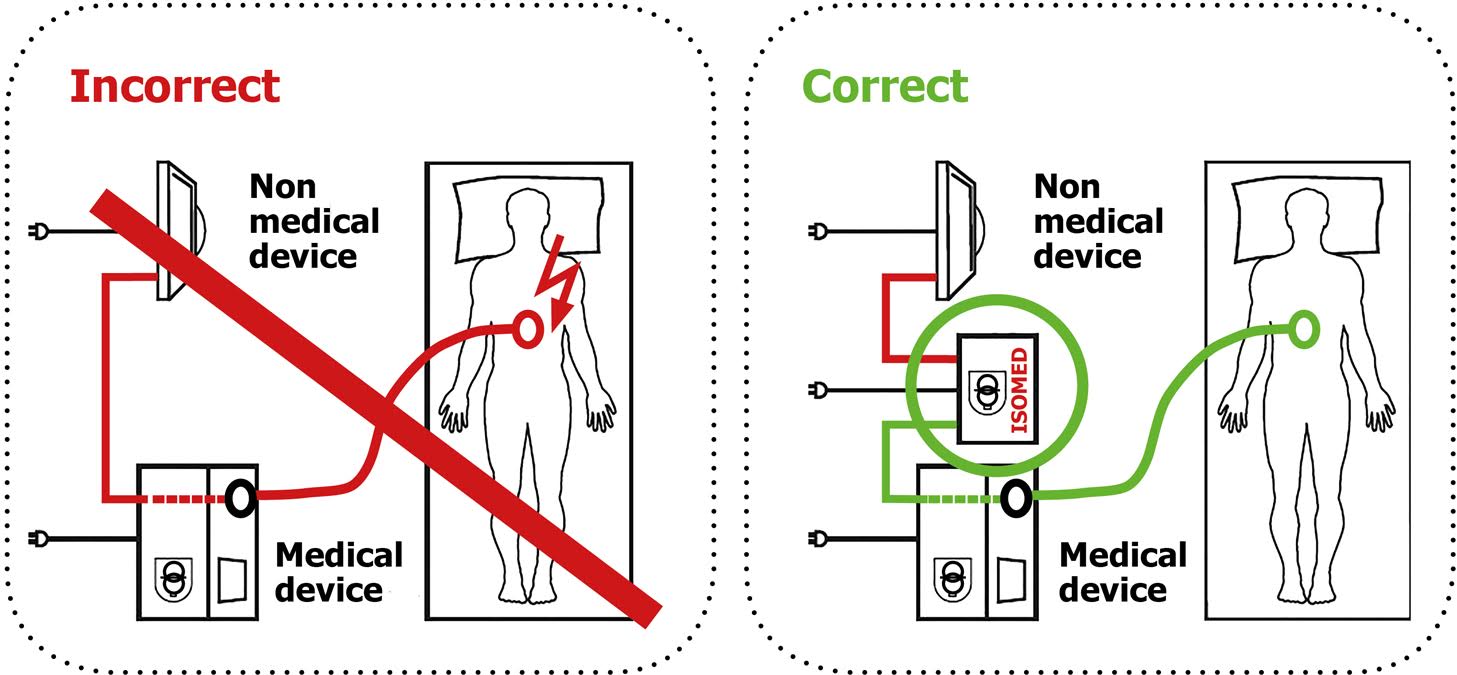
Connecting medical devices such as ultrasound or endoscopy devices to printers, monitors or storage media which are not subject to the stringent standards for medical devices voids their operating licence.
The Medical Devices Directive and the German Medical Device Operating Ordinance (MPBetreibV) stipulate a galvanic isolation for this in order to protect patients and operators against fatal leakage currents caused by defective periphery devices.
The European standard EN 60601-1 for medical electrical devices additionally requires an approved safety transformer and the required creepage distance of 8 mm for a dielectric strength of 4,000 V between power supply and inputs/outputs. This is the only way of ensuring safe operation of the medical devices together with the corresponding non-medical devices.
The ISOMED is a class I medical device and complies with the requirements of the directive 93/42/EEC and EN 60601-1. It is consequently a cost-effective way of implementing the legal requirements.